An integrated in planta infection and in vitro selection strategy for Agrobacterium-mediated transformation of Tagetes erecta
DOI:
https://doi.org/10.31849/29079Keywords:
genetic transformation, hygromycin selection , ornamental plant biotechnology, seed transformation, plant regenerationAbstract
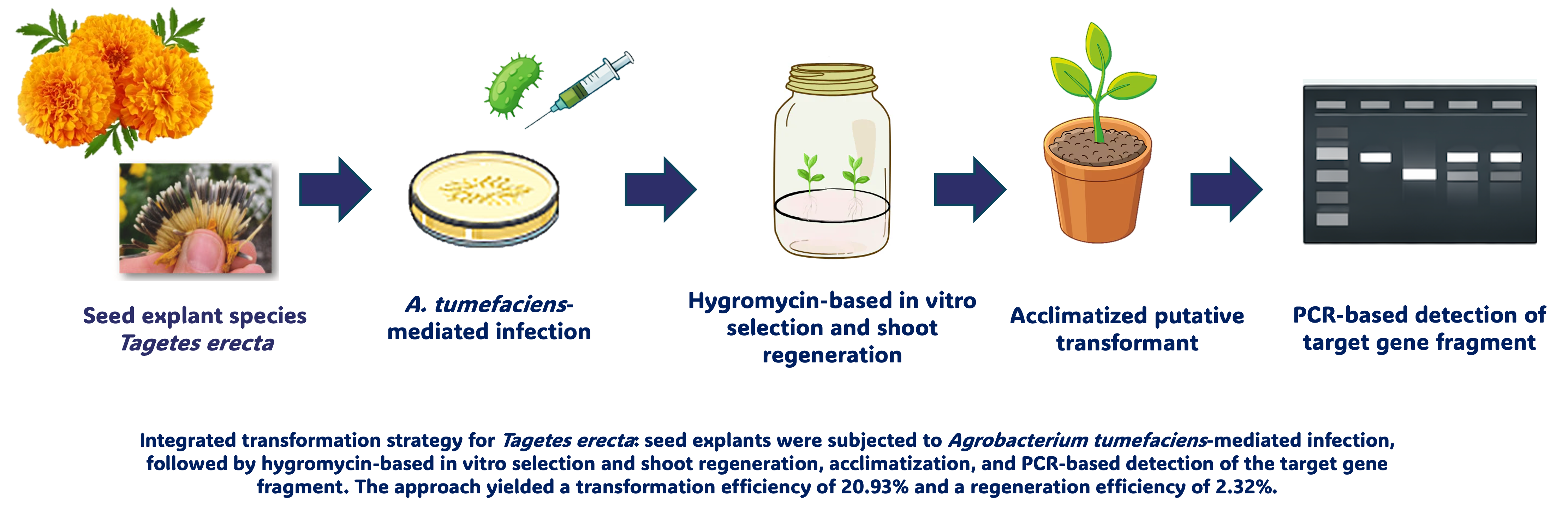
Marigold (Tagetes erecta) is an important ornamental plant valued for its diverse flower colors and morphological variation. Enhancing the genetic variability of this species can be achieved through biotechnological approaches, particularly genetic transformation mediated by Agrobacterium tumefaciens. This study aimed to evaluate an integrated transformation strategy that combines in planta infection with in vitro selection and regeneration in T. erecta. Gene transfer was initiated through in planta infection, followed by hygromycin-based selection and shoot regeneration under in vitro conditions. This approach resulted in a transformation efficiency of 20.93% and a regeneration efficiency of 2.32%, calculated based on the number of hygromycin-resistant explants and regenerated shoots relative to the total number of treated seeds. PCR-based molecular analysis detected the presence of the target gene fragment in putative transformants, suggesting that gene transfer events may have occurred during the transformation process. However, PCR detection alone cannot confirm stable genomic integration or inheritance of the introduced gene. Despite the relatively low regeneration efficiency, the integration of in planta transformation with in vitro selection demonstrates potential as a preliminary strategy for further optimization of transformation protocols in T. erecta, and may support the development of more practical transformation approaches in ornamental plant breeding.
References
Amal, T., Karthika, P., Dhandapani, G., Selvakumar, S., & Vasanth, K. (2020). A simple and efficient Agrobacterium-mediated in planta transformation protocol for horse gram (Macrotyloma uniflorum Lam. Verdc.). Journal of Genetic Engineering and Biotechnology, 18, Article 23. https://doi.org/10.1186/s43141-020-00023-z
Anjelita, D., Purnama, I., Lidar, S., Mutamima, A., & Susanti, Y. (2025). Cloche duration and auxin-based regulators using standardised indole-3-acetic acid for growth dynamics of rose (Rosa chinensis) cuttings. Journal of Agriculture and Rural Development in the Tropics and Subtropics, 126(1), 141–150. https://doi.org/10.17170/kobra-2025052211169
Azizi-Dargahlou, S., & Pouresmaeil, M. (2024). Agrobacterium tumefaciens-mediated plant transformation: A review. Molecular Biotechnology, 66, 1563–1580. https://doi.org/10.1007/s12033-023-00788-x
Beti, J. A. (2020). Marigold (Tagetes erecta L.) tanaman hias potensial multiguna. In Prosiding Seminar Nasional Pertanian Peternakan Terpadu ke-3 (pp. 592–602). http://eproceedings.umpwr.ac.id/index.php/pertanian/article/view/1301
Bhuyan, S., Kumar, M., Devde, P., Rai, A., Mishra, A., Singh, P., & Siddique, K. (2023). Progress in gene editing tools, implications and success in plants: A review. Frontiers in Genome Editing, 5, 1272678. https://doi.org/10.3389/fgeed.2023.1272678
Doyle, J. J., & Doyle, J. L. (1987). A rapid DNA isolation procedure for small quantities of fresh leaf tissue. Phytochemical Bulletin, 19(1), 11–15.
Gelvin, S. B. (2021). Plant DNA repair and Agrobacterium T-DNA integration. International Journal of Molecular Sciences, 22(16), 8458. https://doi.org/10.3390/ijms22168458
Grzelak, M., Pacholczak, A., & Nowakowska, K. (2024). Challenges and insights in the acclimatization step of micropropagated woody plants. Plant Cell, Tissue and Organ Culture, 159, 72. https://doi.org/10.1007/s11240-024-02923-1
Hanana, M., Ayadi, R., Mzid, R., Khouja, M. L., Hanachi, A. S., & Hamrouni, L. (2018). Efficient method of seed transformation via Agrobacterium tumefaciens for obtaining transgenic plants of Hibiscus cannabinus L. Industrial Crops and Products, 113, 274–282. https://doi.org/10.1016/j.indcrop.2018.01.050
Karjee, S. (2017). Development of in vitro protocol for efficient regeneration of marigold (Tagetes sp.) using non-axillary explants (Master’s thesis). Indian Agricultural Research Institute.
Karthik, S., Pavan, G., Sathish, S., Siva, R., Kumar, P. S., & Manickavasagam, M. (2018). Genotype-independent and enhanced in planta Agrobacterium tumefaciens-mediated genetic transformation of peanut (Arachis hypogaea L.). 3 Biotech, 8(4), 201. https://doi.org/10.1007/s13205-018-1231-1
Lenawaty, D. Y., Sukma, D., Syukur, M., Suprapta, D. N., Nurcholis, W., & Aisyah, S. I. (2022). Increasing the diversity of marigold (Tagetes sp.) by acute and chronic chemical induced mutation of EMS (ethyl methane sulfonate). Biodiversitas, 23(3), 1399–1407. https://doi.org/10.13057/biodiv/d230326
Mei, G., Chen, A., Wang, Y., Li, S., Wu, M., Hu, Y., Liu, X., & Hou, X. (2024). A simple and efficient in planta transformation method based on the active regeneration capacity of plants. Plant Communications, 5, 100822. https://doi.org/10.1016/j.xplc.2024.100822
Moniruzzaman, M., Zhong, Y., Huang, Z., Yan, H., Yuanda, L., Jiang, B., & Zhong, G. (2021). Citrus cell suspension culture establishment, maintenance, efficient transformation and regeneration to complete transgenic plant. Plants, 10(4), 664. https://doi.org/10.3390/plants10040664
Narushima, M., Uesugi, M., Murai, Y., Katayama, Y., Iimura, Y., & Kajita, S. (2017). In vitro regeneration and Agrobacterium-mediated transformation of male-sterile marigold (Tagetes erecta L.). Plant Biotechnology, 34, 125–129. https://doi.org/10.5511/plantbiotechnology.17.0530a
Ontiveros-Cisneros, A., Moss, O., Van Moerkercke, A., & Van Aken, O. (2022). Evaluation of antibiotic-based selection methods for Camelina sativa stable transformants. Cells, 11, 1068. https://doi.org/10.3390/cells11071068
Patra, A., Samal, K., Rout, G., Sahu, S., & Jagadev, P. (2021). Agrobacterium tumefaciens-mediated genetic transformation of green gram (Vigna radiata L.). Legume Research. https://doi.org/10.18805/lr-4643
Prem Kumar, G., Sivakumar, S., Siva, G., Vigneswaran, M., Senthil Kumar, T., & Jayabalan, N. (2021). Optimization and establishment of genotype-independent seed-based in planta transformation system in cotton (Gossypium hirsutum L.). Plant Gene, 27, 100296. https://doi.org/10.1016/j.plgene.2021.100296
Sanjawani, W. (2017). Potensi tinggi, market bunga gumitir capai 200 milyar setahun. https://www.balipost.com/news/2017/06/01/10266
Sisharmini, A., Purwoko, B. S., Khumaida, N., & Trijatmiko, K. R. (2018). Optimization of acetosyringone and hygromycin concentrations in genetic transformation of rice cv. Fatmawati mediated by Agrobacterium tumefaciens. Indonesian Journal of Agricultural Science, 46(3), 223–230.
Sjahril, R., Jamaluddin, I., Nadir, M., Asman, & Dungga, N. E. (2018). Effect of selection agents on chrysanthemum (Chrysanthemum morifolium) callus growth after Agrobacterium-mediated genetic transformation. IOP Conference Series: Earth and Environmental Science, 157, 012044. https://doi.org/10.1088/1755-1315/157/1/012044
Wang, P., Si, H., Li, C., Xu, Z., Guo, H., Jin, S., & Cheng, H. (2025). Plant genetic transformation: Achievements, current status and future prospects. Plant Biotechnology Journal, 23, 2034–2058. https://doi.org/10.1111/pbi.70028
Zhang, Y., Zhang, D., Zhong, Y., Chang, X., Hu, M., & Cheng, C. (2017). A simple and efficient in planta transformation method for pommelo (Citrus maxima) using Agrobacterium tumefaciens. Scientia Horticulturae, 214, 174–179. https://doi.org/10.1016/j.scienta.2016.11.033
Zhang, H., Xin, H., Cong, R., Li, Z., Song, L., & Wei, Z. (2019). Cross compatibility analysis to identify suitable parents of Tagetes erecta and T. patula for heterotic hybrid breeding. Notulae Botanicae Horti Agrobotanici Cluj-Napoca, 47(3), 676–682. https://doi.org/10.15835/nbha47311388
Downloads
Published
Issue
Section
License
Copyright (c) 2026 Universitas Lancang Kuning

This work is licensed under a Creative Commons Attribution 4.0 International License.













