Physiological responses of pakcoy (Brassica rapa ssp. chinensis) to chitosan application under short-term drought stress
DOI:
https://doi.org/10.31849/29973Keywords:
chitosan biostimulant, oxidative stress, proline accumulation, water deficit, principal component analysisAbstract
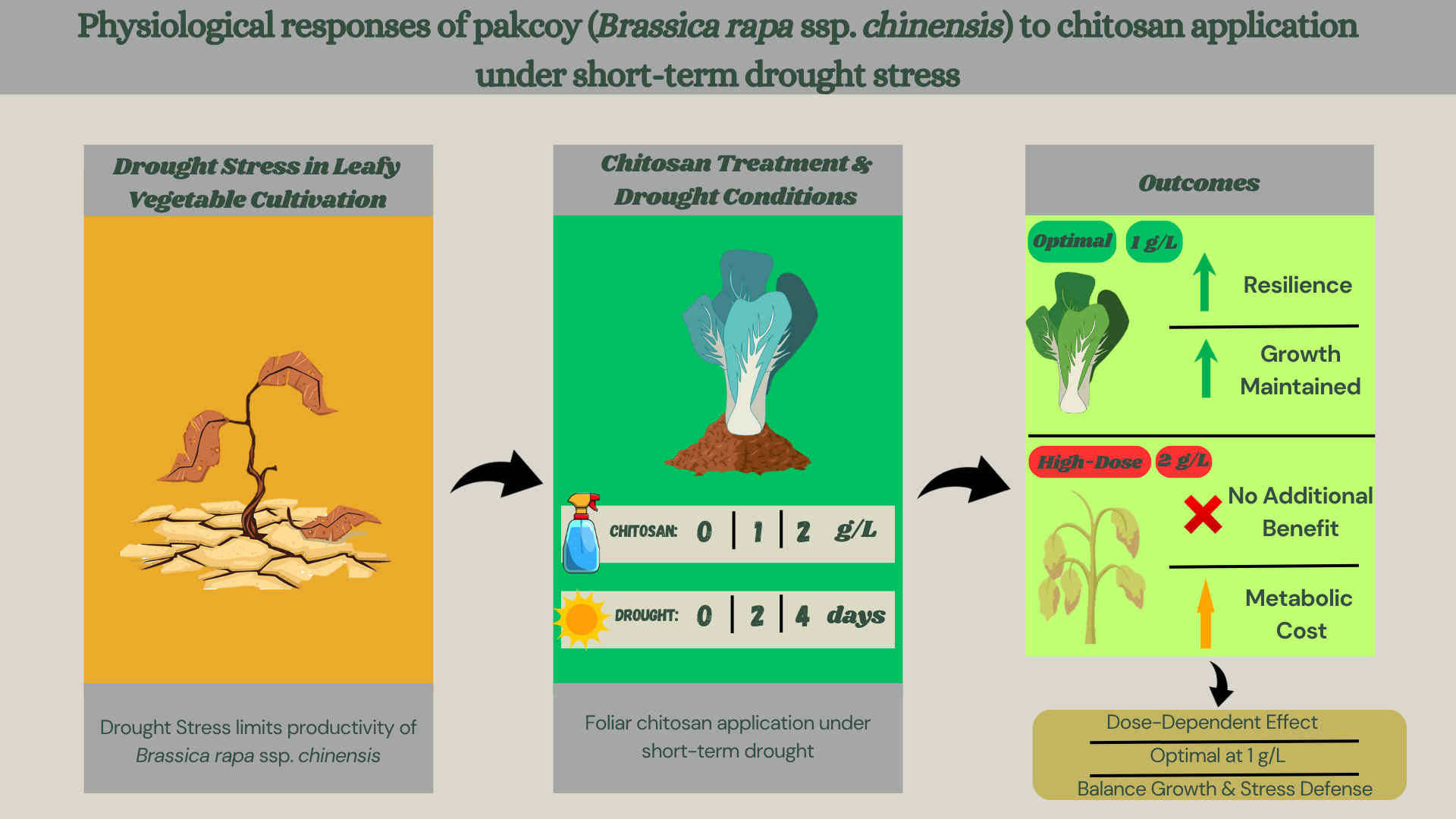
Drought stress is a major environmental factor limiting leafy vegetable productivity in greenhouse cultivation. Chitosan is widely reported as a biostimulant that may enhance stress tolerance through physiological and biochemical regulation. This study aimed to evaluate physiological responses of pakcoy (Brassica rapa ssp. chinensis) to foliar-applied chitosan under short-term drought stress conditions. The experiment was conducted in a greenhouse using a factorial design with three chitosan concentrations (0, 1, and 2 g L⁻¹) and three drought durations (0, 2, and 4 days without irrigation prior to harvest). Growth parameters, photosynthetic pigments, proline accumulation, hydrogen peroxide (H₂O₂) content, and leaf area were measured to assess plant responses. Results showed that moderate chitosan application (1 g L⁻¹) helped maintain plant growth and physiological stability under mild drought conditions. In contrast, higher chitosan concentrations increased oxidative responses, indicated by elevated H₂O₂ and proline levels. Principal component analysis revealed distinct physiological response patterns associated with chitosan dosage and drought duration. The study provides insights into the dose-dependent role of chitosan in modulating physiological responses of leafy vegetables under short-term water deficit.
References
Abdelaal, K., Attia, K. A., Niedbała, G., Wojciechowski, T., Hafez, Y., Alamery, S., Alateeq, T. K., & Arafa, S. A. (2021). Mitigation of drought damages by exogenous chitosan and yeast extract with modulating the photosynthetic pigments, antioxidant defense system and improving the productivity of garlic plants. Horticulturae, 7(11), 1–17. https://doi.org/10.3390/horticulturae7110510
Abobatta, W. F. (2023). Chitosan: A promising plant stimulant. International Journal of Agricultural Science and Food Technology, 9(4), 98–103. https://doi.org/10.17352/2455-815X.000199
Aranaz, I., Alcántara, A. R., Civera, M. C., Arias, C., Elorza, B., Heras Caballero, A., & Acosta, N. (2021). Chitosan: An overview of its properties and applications. Polymers, 13(19), 1–27. https://doi.org/10.3390/polym13193256
Ávila, R. G., Magalhães, P. C., Vitorino, L. C., Bessa, L. A., de Souza, K. R. D., Queiroz, R. B., Jakelaitis, A., & Teixeira, M. B. (2023). Chitosan induces sorghum tolerance to water deficits by positively regulating photosynthesis and the production of primary metabolites, osmoregulators, and antioxidants. Journal of Soil Science and Plant Nutrition, 23(1), 1156–1172. https://doi.org/10.1007/s42729-022-01111-4
Bates, L. S., Waldren, R. P., & Teare, I. D. (1973). Rapid determination of free proline for water-stress studies. Plant and Soil, 39(1), 205–207. https://doi.org/10.1007/BF00018060
Bayram, D. D., Yigit, E., & Akbulut, G. B. (2015). The effects of salicylic acid on Helianthus annuus L. exposed to quizalofop-p-ethyl. American Journal of Plant Sciences, 6(14), 2412–2425. https://doi.org/10.4236/ajps.2015.614244
Berliana, A. I., Kuswandari, C. D., Retmana, B. P., Putrika, A., & Purbaningsih, S. (2020). Analysis of the potential application of chitosan to improve vegetative growth and reduce transpiration rate in Amaranthus hybridus. IOP Conference Series: Earth and Environmental Science, 481(1), 1–6. https://doi.org/10.1088/1755-1315/481/1/012021
Bistgani, Z. E., Siadat, S. A., Bakhshandeh, A., Pirbalouti, A. G., & Hashemi, M. (2017). Interactive effects of drought stress and chitosan application on physiological characteristics and essential oil yield of Thymus daenensis Celak. The Crop Journal, 5(5), 407–415. https://doi.org/10.1016/j.cj.2017.04.003
Castro-Valdecantos, P., Martínez-Melgarejo, P. A., Pérez-Alfocea, F., Tian, J., & Dodd, I. C. (2023). Stem girdling enhances ABA-induced stomatal closure of phosphorus-deprived soybean plants. Environmental and Experimental Botany, 159, 149–156. https://doi.org/10.1016/j.envexpbot.2023.105266
Dao, J., Xing, Y., Chen, C., Chen, M., Wang, Z., & Chen, Y. (2025). Changes in shoot and root adaptations of fibrous-root and taproot crops in response to different drought types: A meta-analysis. Agricultural Water Management, 309, 1–10. https://doi.org/10.1016/j.agwat.2025.109320
Franco‐Navarro, J. D., Padilla, Y. G., Álvarez, S., Calatayud, Á., Colmenero‐Flores, J. M., Gómez‐Bellot, M. J., Hernández, J. A., Martínez‐Alcalá, I., Penella, C., Pérez‐Pérez, J. G., Sánchez‐Blanco, M. J., Tasa, M., & Acosta‐Motos, J. R. (2025). Advancements in water‐saving strategies and crop adaptation to drought: A comprehensive review. Physiologia Plantarum, 177(4), 1–49. https://doi.org/10.1111/ppl.70332
García-García, A. L., Matos, A. R., Feijão, E., Cruz de Carvalho, R., Boto, A., Marques da Silva, J., & Jiménez-Arias, D. (2023). The use of chitosan oligosaccharide to improve artemisinin yield in well-watered and drought-stressed plants. Frontiers in Plant Science, 14, 1–17. https://doi.org/10.3389/fpls.2023.1200898
Goñi, M. G., Tomadoni, B., Roura, S. I., & Moreira, M. del R. (2017). Lactic acid as potential substitute of acetic acid for dissolution of chitosan: preharvest application to Butterhead lettuce. Journal of Food Science and Technology, 54(3), 620–626. https://doi.org/10.1007/s13197-016-2484-5
Haghaninia, M., Rasouli, F., Javanmard, A., Mahdavinia, G., Azizi, S., Nicoletti, R., Murariu, O. C., Tallarita, A. V., & Caruso, G. (2024). Improvement of physiological features and essential oil content of Thymus vulgaris after soil amendment with chitosan nanoparticles under chromium toxicity. Horticulturae, 10(6), 1–24. https://doi.org/10.3390/horticulturae10060659
Haghpanah, M., Hashemipetroudi, S., Arzani, A., & Araniti, F. (2024). Drought tolerance in plants: Physiological and molecular responses. Plants, 13(21), 1–30. https://doi.org/10.3390/plants13212962
Hidangmayum, A., Dwivedi, P., Katiyar, D., & Hemantaranjan, A. (2019). Application of chitosan on plant responses with special reference to abiotic stress. Physiology and Molecular Biology of Plants, 25(2), 313–326. https://doi.org/10.1007/s12298-018-0633-1
Jana, S., & Choudhuri, M. A. (1982). Glycolate metabolism of three submersed aquatic angiosperms during ageing. Aquatic Botany, 12, 345–354. https://doi.org/10.1016/0304-3770(82)90026-2
Jeon, J., Lim, C. J., Kim, J. K., & Park, S. U. (2018). Comparative metabolic profiling of green and purple pakchoi (Brassica rapa subsp. chinensis). Molecules, 23(7), 1–11. https://doi.org/10.3390/molecules23071613
Ji, H., Wang, J., Chen, F., Fan, N., Wang, X., Xiao, Z., & Wang, Z. (2022). Meta-analysis of chitosan-mediated effects on plant defense against oxidative stress. Science of The Total Environment, 851(1), 158212. https://doi.org/10.1016/j.scitotenv.2022.158212
Jia, J., Zhao, M., Liu, R., Xue, C., Xia, Z., Hu, B., & Rennenberg, H. (2024). Drought-mediated oxidative stress and its scavenging differ between citrus hybrids with medium and late fruit maturation. Plant Stress, 14, 1–16. https://doi.org/10.1016/j.stress.2024.100670
Juan, C. A., Pérez de la Lastra, J. M., Plou, F. J., & Pérez-Lebeña, E. (2021). The chemistry of reactive oxygen species (ROS) revisited: Outlining their role in biological macromolecules (DNA, lipids and Proteins) and induced pathologies. International Journal of Molecular Sciences, 22(9), 1–21. https://doi.org/10.3390/ijms22094642
Kaur, G., & Asthir, B. (2017). Molecular responses to drought stress in plants. Biologia Plantarum, 61(2), 201–209. https://doi.org/10.1007/s10535-016-0700-9
Ke, X., Yao, J., Jiang, Z., Gu, X., & Xu, P. (2025). Recover and surpass: The mechanisms of plants transition upon rehydration from drought. Plant Stress, 15, 1–10. https://doi.org/10.1016/j.stress.2025.100782
Khalil, H. A., & Badr Eldin, R. M. (2021). Chitosan improves morphological and physiological attributes of grapevines under deficit irrigation conditions. Journal of Horticultural Research, 29(1), 9–22. https://doi.org/10.2478/johr-2021-0003
Lichtenthaler, H. K., & Wellburn, A. R. (1983). Determinations of total carotenoids and chlorophylls a and b of leaf extracts in different solvents. Biochemical Society Transactions, 11(5), 591–592. https://doi.org/10.1042/bst0110591
Malekpoor, F., Pirbalouti, A. G., & Salimi, A. (2016). Effect of foliar application of chitosan on morphological and physiological characteristics of basil under reduced irrigation. Research on Crops, 17(2), 354–359. https://doi.org/10.5958/2348-7542.2016.00060.7
Mueangnak, K., Kitwetcharoen, H., Thanonkeo, S., Klanrit, P., Apiraksakorn, J., Klanrit, P., Klanrit, P., & Thanonkeo, P. (2025). Enhancing betalains production and antioxidant activity in Celosia argentea cell suspension cultures using biotic and abiotic elicitors. Scientific Reports, 15(1), 1–17. https://doi.org/10.1038/s41598-024-83096-x
Nasiri, E. (2024). Nutritional and bioactive compounds of Bok-choy: Beneficial effects on human health. Journal of Natural Science Review, 2(Special.Issue), 148–163. https://doi.org/10.62810/jnsr.v2iSpecial.Issue.122
Nimnuan, S., Piriyaphattarakit, A., Hanpattanakit, P., Jampasri, K., & Saeng-ngam, S. (2025). Effects of short-term drought stress on chlorophyll fluorescence and proline content of Ficus annulata. Journal of Current Science and Technology, 16(1), 1–14. https://doi.org/10.59796/jcst.V16N1.2026.150
Ort, D. R., Merchant, S. S., Alric, J., Barkan, A., Blankenship, R. E., Bock, R., Croce, R., Hanson, M. R., Hibberd, J. M., Long, S. P., Moore, T. A., Moroney, J., Niyogi, K. K., Parry, M. A. J., Peralta-Yahya, P. P., Prince, R. C., Redding, K. E., Spalding, M. H., van Wijk, K. J., Vermaas, W. F. J., von Caemmerer, S., Weber, A. P. M., Yeates, T. O., Yuan, J. S., & Zhu, X. G. (2015). Redesigning photosynthesis to sustainably meet global food and bioenergy demand. Proceedings of the National Academy of Sciences, 112(28), 8529–8536. https://doi.org/10.1073/pnas.1424031112
Park, J.-E., Kim, J., Purevdorj, E., Son, Y.-J., Nho, C. W., & Yoo, G. (2021). Effects of long light exposure and drought stress on plant growth and glucosinolate production in pak choi (Brassica rapa subsp. chinensis). Food Chemistry, 340, 1–8. https://doi.org/10.1016/j.foodchem.2020.128167
Pirbalouti, A. G., Malekpoor, F., Salimi, A., & Golparvar, A. (2017). Exogenous application of chitosan on biochemical and physiological characteristics, phenolic content and antioxidant activity of two species of basil (Ocimum ciliatum and Ocimum basilicum) under reduced irrigation. Scientia Horticulturae, 217, 114–122. https://doi.org/10.1016/j.scienta.2017.01.031
Rao, M. J., Duan, M., Zhou, C., Jiao, J., Cheng, P., Yang, L., Wei, W., Shen, Q., Ji, P., Yang, Y., Conteh, O., Yan, D., Yuan, H., Rauf, A., Ai, J., & Zheng, B. (2025). Antioxidant defense system in plants: Reactive oxygen species production, signaling, and scavenging during abiotic stress-induced oxidative damage. Horticulturae, 11(5), 1–33. https://doi.org/10.3390/horticulturae11050477
Seleiman, M. F., Al-Suhaibani, N., Ali, N., Akmal, M., Alotaibi, M., Refay, Y., Dindaroglu, T., Abdul-Wajid, H. H., & Battaglia, M. L. (2021). Drought stress impacts on plants and different approaches to alleviate its adverse effects. Plants, 10(2), 1–25. https://doi.org/10.3390/plants10020259
Sharif, R., Mujtaba, M., Ur Rahman, M., Shalmani, A., Ahmad, H., Anwar, T., Tianchan, D., & Wang, X. (2018). The multifunctional role of chitosan in horticultural crops; A review. Molecules, 23(4), 1–20. https://doi.org/10.3390/molecules23040872
Taghavi, T., Patel, H., & Rafie, R. (2022). Anthocyanin extraction method and sample preparation affect anthocyanin yield of strawberries. Natural Product Communications, 17(5), 1–7. https://doi.org/10.1177/1934578X221099970
Yang, X., Lu, M., Wang, Y., Wang, Y., Liu, Z., & Chen, S. (2021). Response mechanism of plants to drought stress. Horticulturae, 7(3), 1–36. https://doi.org/10.3390/horticulturae7030050
Zia, R., Nawaz, M. S., Siddique, M. J., Hakim, S., & Imran, A. (2021). Plant survival under drought stress: Implications, adaptive responses, and integrated rhizosphere management strategy for stress mitigation. Microbiological Research, 242, 1–16. https://doi.org/10.1016/j.micres.2020.126626
Zong, H., Liu, S., Xing, R., Chen, X., & Li, P. (2017). Protective effect of chitosan on photosynthesis and antioxidative defense system in edible rape (Brassica rapa L.) in the presence of cadmium. Ecotoxicology and Environmental Safety, 138, 271–278. https://doi.org/10.1016/j.ecoenv.2017.01.009
Downloads
Additional Files
Published
Issue
Section
License
Copyright (c) 2026 Universitas Lancang Kuning

This work is licensed under a Creative Commons Attribution 4.0 International License.













